A new project is underway at the KIOS Research Center researching a break-through solution for improved management of colorectal cancer and potentially other malignancies whose cells express specific molecules which belong to the epidermal growth factor family of receptors (EGFR). This research project with the acronym FWTAYGEIA (pronounced photagvia – meaning luminiscence ) combines the expertise of various fields to develop a novel group of pharmaceuticals which have both therapeutic and diagnostic qualities.
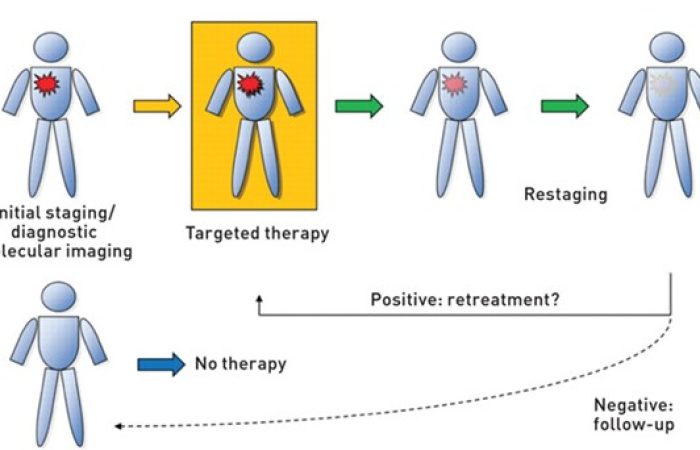
These types of pharmaceuticals, which are categorized as “theranostic”, have the ability to target cancer cells, enable both imaging of the malignancy (diagnosis) whilst simultaneously having a therapeutic effect (therapy). The ability of the envisioned “theranostic agents” to quantitatively and non-invasively image well-defined molecular targets on cancer cells is expected to improve lesion detection, patient stratification, drug validation, dose optimization, delineation of surgical margins, and treatment monitoring. These characteristics can lead to improved prognosis due to earlier detection. Cancer will be detected in early molecular and biochemical stages, before pathological changes become visible. These novel agents will be capable of performing multi-modal imaging, using both MRI and near infrared (NIR) optical fluorescence endoscopy. The agents will uniquely combine the abilities of whole-body imaging performed by an MIR with the high resolution, with cellular level, surveillance provided usually by endoscopy.
In addition, the theranostic agent will enable the targeting of specific markers of the disease, allowing for earlier and targeted therapy, better treatment, reduced side-effects, and better prognosis. It will enable individualized cancer patient management, ultimately improving quality of life, and will offer an efficient and a cost-effective solution to the management of healthcare provision.
The project brings together expertise in medical optics and optoelectronics, magnetics, coordinate Lanthanide chemistry, bio-organic synthesis, and molecular oncology.
The research will focus on enabling the quantitative imaging of the EGFR-related processes, in a non-invasive manner. EGFR is the human epidermal growth factor receptor family that controls a complex network of cellular interactions and responses. Disruptions in this function lead to various malignancies, including colorectal cancer. Quantitative imaging of EGFR can aid in lesion detection, patient stratification, new drug development and validation, dose optimization and treatment monitoring.
The approach will focus on Near Infrared (NIR) emitting probes, which are mostly preferred for biological applications due to (a) lack of native NIR fluorescence in biological systems leading to highly sensitive detection, delineating molecular structures at pico molar concentrations (i.e. ~ 10-7 mg/l), (b) low scattering of NIR photons allowing for better image resolution, and (c) adequate tissue penetration permitting non-invasive or minimally invasive imaging with the aid of an endoscope.
Unfortunately, optical imaging is inadequate in surveying the entire body for metastasis, a function for which MRI is well-suited. However, MRI also has significant limitations as the inherent low sensitivity of the MR probes make validation of in vivo imaging experiments by more than one approach essential. This problem cannot be solved by merely adding two different classes of probes, e.g. optical and MRI together, unless they have identical pharmacokinetic properties. Further, optical imaging with anti-EGFR antibodies failed to introduce a therapeutic functionality. Thus, multimodal probes are needed in order to create scaffolds for the amplification of MRI contrast, enable sensitive NIR optical imaging, and provide a significant therapeutic effect. This project proposes the combination of optical with magnetic properties within the same stable molecular structure also possessing anti-EGFR therapeutic potential, with optimized in vivo stability, targeting efficacy and desirable pharmacokinetics for clinical translation.
The research is being led by Professor Costas Pitris at the Biomedical Imaging & Applied Optics Laboratory of the KIOS Research Center.





This project is funded by the Cyprus Research Promotion Foundation through the Framework Programme for Research, Technological Development and Innovation 2009-10 (DESMI 2009-2010), co-funded by the Republic of Cyprus and the European Regional Development Fund.